BD Veritor™ System
A RAPID ONE-STEP ANTIGEN TESTThe BD Veritor System for Rapid Detection of SARS-CoV-2 is a chromatographic immunoassay for the direct and qualitative detection of SARS-CoV-2 antigens in nasal swabs from patients with signs and symptoms who are suspected of COVID-19.
DESCRIPTION
The Antigen Testing System diagnostic tests quickly detect fragments of proteins found on or within the virus by testing samples collected from the nasal cavity using swabs. This test is authorized for use in high and moderate complexity laboratories certified by Clinical Laboratory Improvement Amendments (CLIA), as well as for point-of-care testing by facilities operating under a CLIA Certificate of Waiver.
Antigen tests are important in the overall response against COVID-19 as they can generally be produced at a lower cost than PCR tests and once multiple manufacturers enter the market, can potentially scale to test millions of Americans per day due to their simpler design, helping our country better identify infection rates closer to real time.
-

- Features and Benefits
- Simplified testing process
- Fast, reliable results
- Workflow efficiency
SPECIFICATION
- Positive Agreement Rate: 84%
- Negative Agreement Rate: 100%
- Portable onsite processing
- Approved for healthcare setting
- Nasal Swab
- 15 Min Processing Time
- Detects active infection
- Throughput = ~96 tests/24 hours
- Less accurate than PCR Testing
- Slower for bulk testing
Related Products
-
LOOKING FOR RESULTS?
Tiger Testing does not provide test results over the phone, email or tawk chat to individuals. If you were tested in Georgia or North Carolina and got a text from Tiger Testing (Radeas Labs) , please call 919-263-1150 or visit https://www.radeas.com/contact-us for test results. This company is not associated with us.


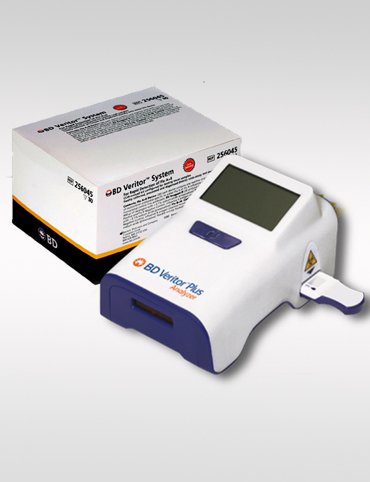
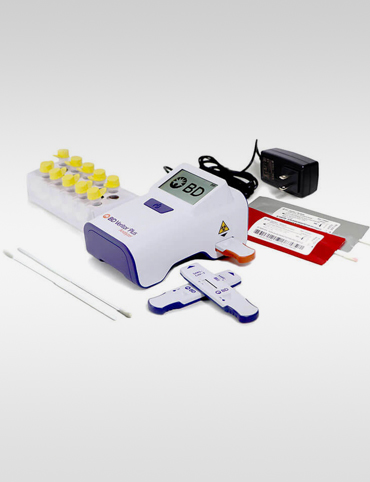






BENEFITS
The portable, easy-to-use BD Veritor™ Plus System provides reliable SARS-CoV-2 results in 15 minutes