Cepheid's GeneXpert™ System
MOLECULAR. FAST AND ACCURATE RESULTS.We all know patients don’t get sick on our schedules. They come to us when they need care — 24 hours a day, 365 days a year. Cepheid’s GeneXpert® System was designed with this in mind — to meet the demands of unpredictable test volumes and deliver fast answers for maximum impact on patient care.
With 20 FDA-cleared tests (including moderate complexity and CLIA waived tests), Cepheid is our partner for empowering clinicians with fast, accurate results.
DESCRIPTION
The GeneXpert Infinity Systems enable a comprehensive menu of tests to be run on a single system, by a single operator, in a moderately complex, real-time environment. With two sizes to choose from, Cepheid’s unique cartridge technology, and exclusive “load and go” capability, the GeneXpert Infinity provides maximum flexibility.
-
-
Key features:
- Adjustable Touch-Screen
- Expanded Conveyor & Loading zone
- Large Test Accumulator Capacity
- Fast, Quiet and Robust Robotic Gantry
- The best things have stayed the same:
Our Patented Cartridge
- Our Patented Module Analytical Unit
- US-IVD. For In Vitro Diagnostic Use.
-

-
NOW AVAILABLE
Cepheid GeneXpert Desktop Systems
With the new addition to our Cepheid family of COVID testing systems, Tiger is able to process more rapidly and efficiently with our new PCR testing systems.
GeneXpert® IV
Dimensions (W x H x D) - 11" x 12" x 11.70"
4 modules (128/day)
-
GeneXpert® XVI
Dimensions (W x H x D) - 22.75" x 25.80" x 13.25"
16 modules (512/day)
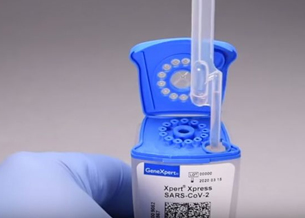
Cepheid has received Emergency Use Authorization (EUA) from the U.S. Food & Drug Administration (FDA) for Xpert® Xpress SARS-CoV-2, a rapid molecular diagnostic test for qualitative detection of SARS-CoV-2, the virus causing COVID-19. The test has been designed to operate on any of Cepheid's more than 23,000 automated GeneXpert® Systems worldwide, with a detection time of approximately 45 minutes.
CEPHEID GENEXPERT INFINITY-48 SPECIFICATION
- Dimensions 216 cm L x 89 cm W x200 cm H
- Weight 1,600 pounds = 725.7 kg
- Throughput >1,300 test per day
Related Products
-
LOOKING FOR RESULTS?
Tiger Testing does not provide test results over the phone, email or tawk chat to individuals. If you were tested in Georgia or North Carolina and got a text from Tiger Testing (Radeas Labs) , please call 919-263-1150 or visit https://www.radeas.com/contact-us for test results. This company is not associated with us.










BENEFITS
Positive results may be detected in as little as 5 minutes
Negative results in 13 minutes
Molecular technology targeting COVID-19 RdRp gene
Designed for near patient testing in a variety of healthcare environments1
Room temperature storage
Direct sample types include: Nasal, Throat, and Nasopharyngeal swabs
Facilitates effective patient management